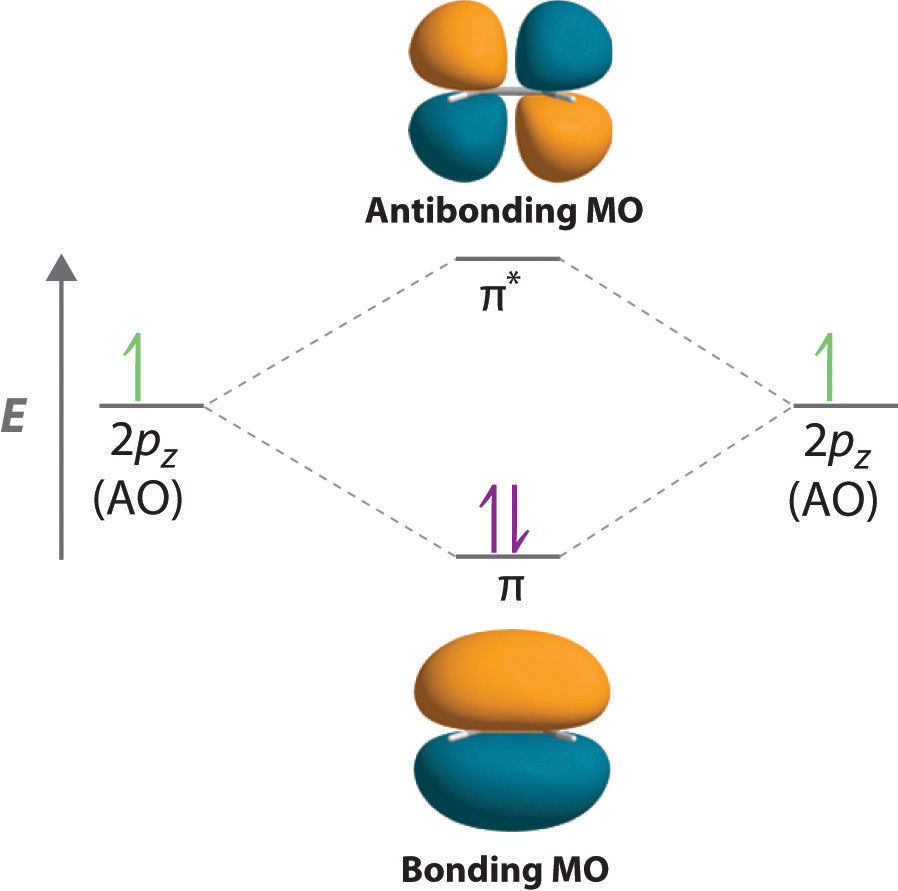
There are two types of molecular orbitals:.This combination is known as the linear combination of atomic orbitals (LCAO). The important point of MOT is that atomic orbitals of different atoms combine to form molecular orbitals.In molecular orbital theory (MOT), the molecular orbitals are formed by the linear combination of atomic orbitals, and the electrons are delocalized over the whole molecule. In valence bond theory (VBT), we deal with the overlapping of atomic orbitals so the electrons are localized between the two atoms. Sigma vs Pi bond by MOT (Molecular Orbital Theory) The VBT can explain the bonding of some simple molecules but in some cases like Be, B, and C, etc, it may not predict the correct results. An electron is present in the 2p z orbital, which corresponds to the pi bond. The hybridized state of oxygen atoms contains two lone pairs, one (half) bond pair which makes a sigma bond. The new hybridized electronic configuration of oxygen atoms is as The ground state electronic configuration of each atom is following: there is no possibility of a pi bond in hydrogen molecules due to limited orbitals.Īccording to VBT, The two oxygen atoms combine to form the oxygen molecule when they are sp 2 hybridized. There exists just a head-on approach, making this bond a sigma bond. The two hydrogen atoms H A and H B each have one electron overlap to form a diatomic molecule H 2. The extent of overlapping decides the strength of bonds.Parallel overlapping (sidewise/lateral).The two types of overlapping are possible:.The bond is formed by the pairing of electrons so the spin of two electrons must be opposite (Pauli’s Exclusion principle).The number of bonds formed by an atom is the same as the number of unpaired electrons in the ground state.The unpaired electrons pair up and the atoms attain a stable configuration like a nearest noble gas.Atoms with unpaired electrons tend to combine with other atoms which also have unpaired electrons.Sigma vs Pi bond by VBT (Valence Bond Theory) A triple bond will contain one sigma and two pi bonds.įor example, all single (covalent) bonds are sigma bonds, like H-P, Na-O, Mg-O, Al-F, etc.įor example, all double bonds like C=C, O=O, etc.įor example, all triple bonds like N≡N, C≡C, C≡N, etc. A double bond contains one sigma and one pi bond. Rules to identify sigma and pi bondsĪ single bond always contains one sigma bond. The greater the bond length, the weaker the bond is.Ī sigma bond is stronger than the pi bond due to the shorter bond length. The strength of the bond depends upon the bond length as well. This phenomenon is due to the increase in electron density when approaching orbitals overlap.Ī sigma bond is stronger than the pi bond due to a greater and stronger overlap of orbitals. The greater the overlapping is, the stronger the bonding will be. The stability of sigma and pi bonds depends upon the extent of 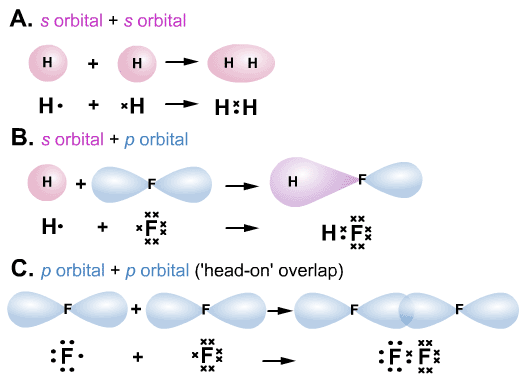
A covalent bond formed by the lateral or side-by-side overlap of half-filled atomic orbitals of atoms is called a pi bond.Sigma bond is the strongest covalent bond formed by head-on overlapping of atomic orbitals.The orbital overlap in the sigma bond is referred to as head to head overlap but for pi bonds, it is called lateral overlap.īoth sigma and pi bonds are actually covalent bonds formed between two atoms. Sigma bond corresponds to the linear or co-axial bond formed between anti-parallel orbitals of two atoms whereas a pi bond is a bond formed between parallel orbitals of two atoms when these orbitals are perpendicular to the sigma bond. Sigma bond Pi bond It is the head-on overlapping result of two atomic orbitals It is the parallel overlapping result of two unhybridized orbitals The linear and co-axial bond formed between two atomic orbitals is termed a sigma bond The sidewise or lateral bond formed between two parallel orbitals is termed a pi bond A stronger covalent bond formed due to transfer of approaching orbitals with heads facing each other A relatively weaker bond formed due to transfer of approaching orbitals with lateral sides facing each other's A sigma bond always occurs as a single bond A pi bond is formed in a compound after a sigma bond has already formed For example, The only bonds between alkanes, etc For example, The bonds between alkenes and alkynes except sigma bond, etc
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
